A vaccine made with messenger RNA (mRNA) technology against glioblastoma, a deadly form of brain cancer, has shown promise in a new clinical trial.
Brain cancer vaccine success in human trials
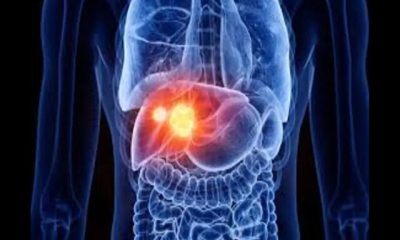
Glioblastoma is one of the deadliest cancers for which few treatment options are available.
Now, a small human clinical trial has shown the efficacy of an mRNA vaccine that rapidly primes the immune system to fight tumors, with promising results, according to NA.
Glioblastoma is the most common form of brain cancer and unfortunately, it is also known as the most aggressive brain cancer.
Glioblastoma is the most common primary malignant tumor of the central nervous system that occurs in the spinal cord or brain. The origin of this tumor is from astrocyte cells (a type of glial cell).
Treatment for glioblastoma includes a combination of surgical removal, radiation therapy, and chemotherapy, but the disease almost always recurs, and patients with it usually survive only about a year after diagnosis, with only about 5 percent of patients surviving more than five years.
A new study from the University of Florida may soon provide these patients with a better option, the mRNA cancer vaccine.
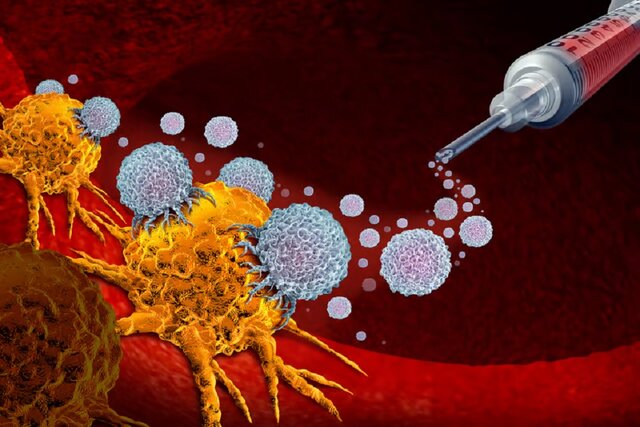
The technology, best known for COVID-19 vaccines, has been shown to quickly prime the immune system to more effectively attack glioblastoma in mice, dogs, and now humans.
As you may remember from the critical days of 2020 and 2021 due to the outbreak of the COVID-19 pandemic, mRNA molecules are essentially natural blueprints that tell cells which proteins to produce and by engineering them to produce harmless versions of the proteins. Associated with pathogens, the immune system can be trained to fight off a true invader when it appears.
Following the real-world success of these treatments during the pandemic, the possibility of adapting mRNA therapies for cancer has emerged with interesting early results.
University of Florida researchers say this new version has two key improvements. First, the vaccine is personalized using samples taken from the patient’s tumor cells. Second, the delivery mechanism is more complex, which ultimately leads to a stronger immune response.
Related article: Testing a vaccine that reduces liver tumors
“Instead of injecting single particles, we inject clusters of particles that are wrapped around each other like the layers of an onion, like a bag full of onions,” said Elias Sayor, senior author of the study. In less than 48 hours, we can see that these tumors change from the so-called cold state, which indicates very few immune cells and a muted immune response, to a warm and highly active immune response state.
He added: This was very surprising. Given how quickly this happened, what it told us is that we were able to activate the early part of the immune system quickly against these cancers, and that’s very important for unlocking the downstream effects of the immune response.
This small clinical trial approved by the US Food and Drug Administration (FDA) was designed to test safety and feasibility and included only four patients with glioblastoma. RNA was extracted from each patient’s tumor after surgical resection, then the mRNA was amplified and wrapped into particle clusters. It was then injected into patients, where it stimulated an immune response.
The team says it’s too early to fully assess the vaccine’s clinical effects, but patients spent more time disease-free and survived longer than expected.
A large-scale trial will be conducted soon, including up to 24 patients, to determine the optimal and safe dose of this vaccine. Next, the next phase of the experiment includes 25 children.
This research was published in the journal Cell.



 Technology9 months ago
Technology9 months ago


 Technology10 months ago
Technology10 months ago


 Technology9 months ago
Technology9 months ago


 Technology11 months ago
Technology11 months ago


 AI1 year ago
AI1 year ago

 Humans1 year ago
Humans1 year ago


 Technology10 months ago
Technology10 months ago

 Technology11 months ago
Technology11 months ago